SEJournal Online is the digital news magazine of the Society of Environmental Journalists. Learn more about SEJournal Online, including submission, subscription and advertising information.
Issue Backgrounder: Primer Helps To Prep for Reporting on PFAS
By Cheryl Hogue
The toxic, persistent compounds known as PFAS are found in increasingly more sources of drinking water. Have they been found tainting drinking water in your town or state?
If not, they probably will be in the future, especially if there’s a military installation or commercial airport or a fire-training facility nearby.
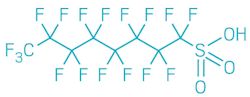 |
| A diagram showing the chemical structure of PFOS, or perfluorooctane sulfonic acid, one of only two PFAS that have been studied extensively and which has been linked to suppression of the immune system and liver effects. Image: Courtesy Chemical & Engineering News. Click to enlarge. |
Some industrial sites are emitting PFAS chemicals into the air or discharging them to water or leaking them into groundwater, rivers, lakes or streams. Just ask the state of Michigan, which has found PFAS pollution near a shuttered paper mill, as well as landfills filled with PFAS-treated shoe leather or auto industry waste.
In many places, these chemicals got into groundwater after military or civilian firefighters released foam in training exercises or as they battled blazes.
Meanwhile, states and EPA are struggling to determine how much PFAS are safe for people to consume in their water. The Interstate Technology & Regulatory Council maintains a series of fact sheets in English and Spanish and keep lists of state actions on these chemicals. The link for this authoritative site is https://pfas-1.itrcweb.org/fact-sheets/.
The PFAS story is expanding fast as researchers are starting to detect these chemicals in food.
So what the heck are PFAS?
PFAS constitute a category of more than 4,000 industrial chemicals that affect the lives of our viewers, listeners and readers nearly every day. Many — but not all — are toxic in ways scientists are still teasing out.
Here’s what journalists need to know as they report on these so-called emerging contaminants.
What is PFAS? The acronym for per- and polyfluoroalkyl substances, a class of chemicals that are persistent or break down into persistent compounds.
Pronunciation note: PFAS as “PEA-fass” and polyfluoroalkyl as “PAUL-ee-floor-oh-AL-kill.”
Style note: As a noun, PFAS is plural, so the “s” is always uppercase. To get around clunky constructions and picky editors, you can use it as an adjective, as in “PFAS pollution.”
Where are PFAS from? PFAS are synthetic chemicals, which means they’re manufactured and don’t occur in nature. Some PFAS are environmental breakdown products of other PFAS.
How many PFAS exist? Estimates from EPA and others are more than 4,000, but no one knows for sure.
What characteristics do all PFAS share? All contain bonds between carbon atoms and fluorine atoms. The carbon-fluorine links are among the strongest chemical bonds in nature and are very hard to break. This characteristic makes PFAS useful — for example, they can withstand high heat — and very persistent in the environment. Some environmental groups call them “forever chemicals” because PFAS don’t naturally break down in time spans that we humans can easily grasp, maybe thousands of years.
Can we get rid of PFAS? Some incineration companies claim their processes destroy nearly 100% of PFAS materials.
What are the two major classes of PFAS?
- The PFAS that are contaminants in water supplies and pose potential health hazards to people and the environment. These are in firefighting foams used to put out oily blazes, such as jet fuel fires. They are also used to make plastic-like PFAS polymers and industries have historically released them into air and water. They are chemically active.
- Plastic-like materials, called polymers, that are in many consumer products like non-stick pans and waterproof raingear. They’re also used in fuel cells. These plastics generally don’t break down and thus are of environmental concern, yet they are essentially inert and don’t pose a toxicity hazard. Be aware that despite some industry claims, scientists are finding that some polymers can break down, creating toxic byproducts.
What are the most (in)famous PFAS?
- The chemical variously called C8, perfluorooctanoic acid (“per-FLOOR-oh-ock-tan-OH-ick acid”) or PFOA (“pea-FO-ah or pea-eff-oh-ay”) was manufactured and used in the United States for decades but no longer is. 3M made it starting in the late 1940s. When 3M stopped producing PFOA in the early 2000s, DuPont began making it but stopped a few years later. Former uses include as an ingredient in firefighting foams and an industrial processing aid in making non-stick coatings.
- Perfluorooctane sulfonic acid (“per-FLOOR-oh-OCK-tain sul-FON-ick acid”) or PFOS (“PEA-fohs or pea-eff-oh-ess”). 3M made this chemical but phased it out in the early 2000s. Former uses: An ingredient in Scotchgard coatings and an ingredient in firefighting foams. Note that both PFOS and PFOA have been found in almost all U.S. residents’ blood. Both chemicals are probably still made elsewhere in the world, notably in Asia.
What about Teflon? Teflon used to be a trademark for DuPont’s plastic-like, non-stick PFAS coating on cookware (a polymer technically called PTFE or polytetrofluoroethylene). Teflon is now a brand name owned by DuPont spinoff Chemours. The company applies it to a swath of PFAS products. They include “fabric protectors” imparting stain-resistance to clothing, paint additives and high-end lubricants for bicycles.
What are the health effects from exposure? Only two PFAS have been studied extensively, PFOA and PFOS. Both are linked to suppression of the immune system and liver effects. The International Agency for Research on Cancer deems PFOA “possibly carcinogenic to humans.” Other PFAS might pose different toxicities. The U.S. Environmental Protection Agency is running tests to determine whether various groups of PFAS sharing particular chemical characteristics might pose similar hazards.
What’s immune toxicity, aka immunotoxicity? A big worry for scientists who study these chemicals is that people, especially children, with high levels of PFAS in their bodies may not react properly to vaccines. That means vaccination may not provide them with immunity to diseases like diphtheria or measles.
Watch for PFAS in the food supply.
The U.S. Department of Agriculture and
the Food and Drug Administration
are starting to probe this issue.
What’s next after drinking water pollution? PFAS in the food supply. A dairy in New Mexico, with wells contaminated by a nearby Air Force base, dumped tainted milk and is euthanizing its cows. A dairy in Maine used sewage sludge biosolids on its fields that was apparently tainted with PFAS — and had to dump its milk too. Australia has warned people living near polluted military bases not to eat milk, eggs or meat from animals raised in the area, or to consume leafy greens from their gardens. The U.S. Department of Agriculture and the Food and Drug Administration are starting to probe this issue.
What units are used for measuring PFAS pollution in water? Parts per trillion. This is the same thing as nanograms per liter (ng/L). A trillion is a one followed by 12 zeros. Be aware that it’s hard for most people to grasp how big a trillion is. A good analogy for one part per trillion? My number-crunching colleagues and I have checked and verified this: It is one drop in 20 filled Olympic-sized swimming pools!
What’s perfluorinated and what’s polyfluorinated? All PFAS are molecules based on a chain of carbon atoms. Perfluorinated means each of the carbon atoms in the chain are bonded to two or three fluorine atoms and no other element. In polyfluorinated molecules, some of the carbons bond with other elements, such as hydrogen and chlorine.
How do you get PFAS out of water? The most widely used technology is granulated activated carbon — GAC (often pronounced “GACK”). Another is reverse osmosis. Both, but especially the latter, pose disposal issues: What happens to all the PFAS removed? Will it go to an incinerator? Will it get disposed of in an underground injection well? Landfilling used, or “spent,” GAC can lead to PFAS contamination of surface or groundwater through leachate. There are other technologies on the horizon as well, including proposed methods to obliterate PFAS in groundwater without pumping to the surface for treatment.
Cheryl Hogue is a reporter for Chemical & Engineering News who has written extensively about PFAS. She was forced to take two years of chemistry as an undergrad biology major and those courses helped her land two journalism jobs. If you are a science nerd who wants to know more about the chemical structures of PFAS chemicals, check out this guide at cenm.ag/pfas.
* From the weekly news magazine SEJournal Online, Vol. 4, No. 25. Content from each new issue of SEJournal Online is available to the public via the SEJournal Online main page. Subscribe to the e-newsletter here. And see past issues of the SEJournal archived here.